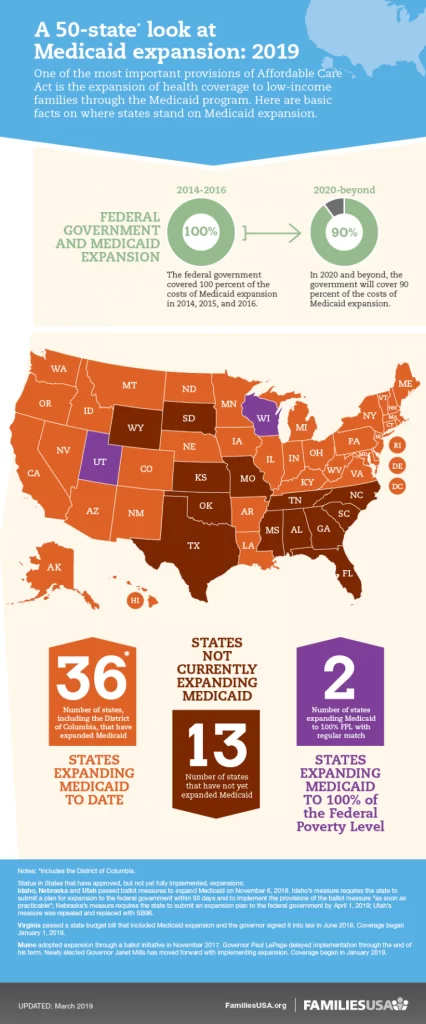
By: Dr. Anish Shah Bronx-Lebanon Hospital; Bronx, NY On March 1, 2024, the U.S. Food and Drug Administration approved amivantamab- vmjw (Rybrevant, Janssen Biotech, Inc.) in combination with carboplatin and pemetrexed for the initial
In this blog article, Dr. Dan Milner, Chief Medical Officer of the American Society for Clinical Pathology, discusses his experience about his participation in the 2022 Summit on Cancer Health Disparities held
MoreThe 2022 Summit on Cancer Health Disparities will bring together oncology professionals, cancer disparity researchers, and other stakeholders to discuss the factors leading to cancer health inequities. Additionally, they will explore ways
MoreFor more information contact: Tara Shah (425) 557-5953 [email protected] Janakpurdham, Nepal, April 9, 2022 – As part of its planned expansion to serve the community of cancer patients in Nepal’s Province 2
MoreThe Binaytara Foundation organized three in-person oncology conferences in October 2021. The 2021 Updates in Hematologic Malignancies and the 2021 Precision Oncology Summit were held in Salt Lake City, Utah on October
More