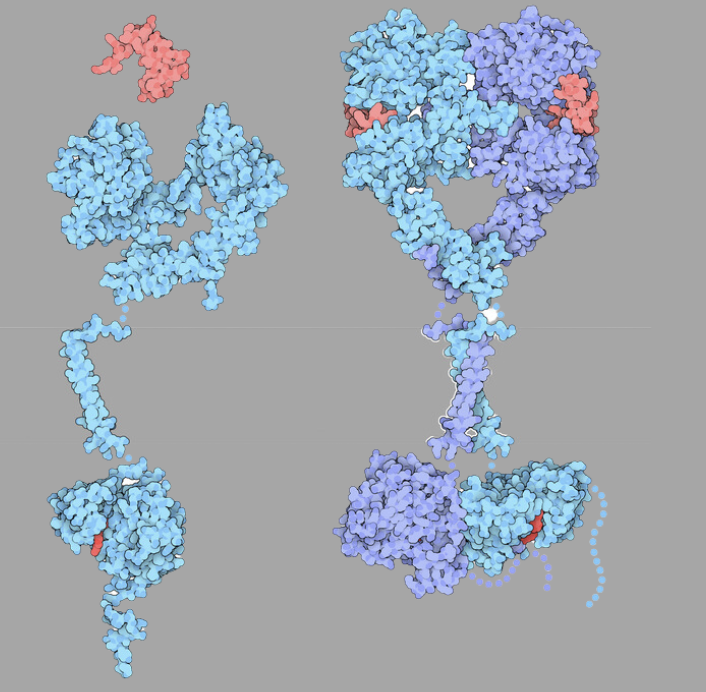
By: Dr. Anish Shah Bronx-Lebanon Hospital; Bronx, NY On March 1, 2024, the U.S. Food and Drug Administration approved amivantamab- vmjw (Rybrevant, Janssen Biotech, Inc.) in combination with carboplatin and pemetrexed for the initial
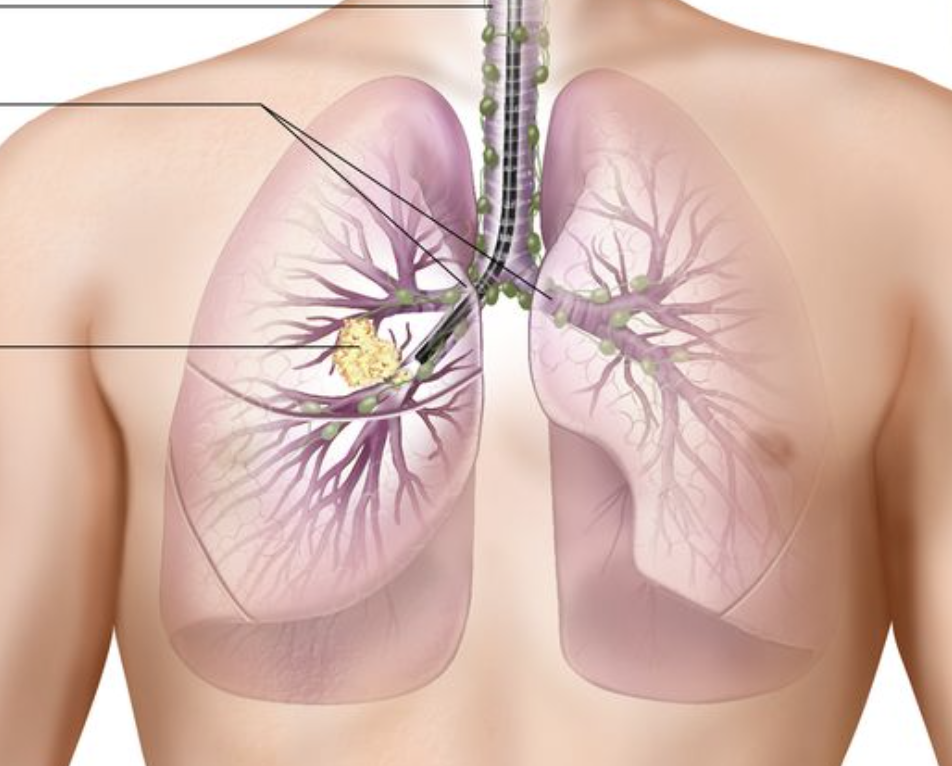
By Dr. Kanak Parmer Texas Tech University Health Sciences Center On October 16,2023 the U.S. Food and Drug Administration approved pembrolizumab for patients with resectable non-small cell lung cancer (NSCLC) tumors
MoreBy Dr. Lei Deng Assistant Professor/Medical Oncologist Thoracic, Head & Neck Medical Oncology University of Washington/Fred Hutchinson Cancer Center https://youtu.be/QVW-vify1x8 Transcript: Dr. Deng: “Hi everyone! Welcome to the Cancer News Book Interview
MoreMARIPOSA-2: A Study of Amivantamab and Lazertinib in Combination With Platinum-Based Chemotherapy Compared With Platinum-Based Chemotherapy in Patients With Epidermal Growth Factor Receptor (EGFR)- Mutated Locally Advanced or Metastatic Non- Small Cell
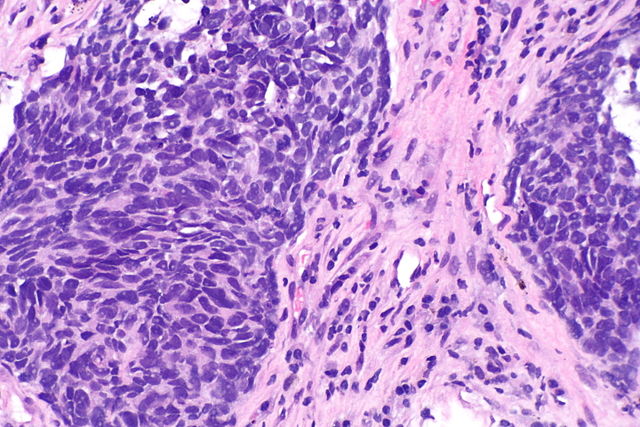
MoreBy Dr. Anam Kamal Ascension Providence Hospital TROPION-Lung01: A randomized, phase-3 study of datopotamab deruxtecan (Dato-DXd; DS-1062) versus docetaxel in previously treated advanced or metastatic non-small cell lung cancer (NSCLC) without actionable
MoreBellevue, WA – October 24, 2024 The Binaytara Foundation, a leading global cancer non-profit organization dedicated to improving access to cancer care, is set to host its annual Global Oncology Summit on
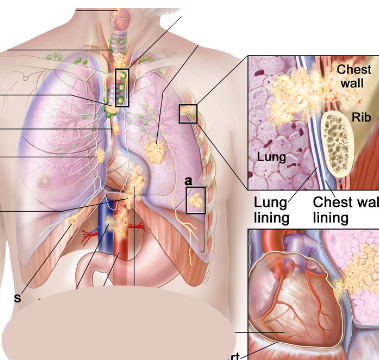
MoreOn October 11, 2023, the U.S. Food and Drug Administration granted approval to Encorafenib (BRAF inhibitor) with Binimetinib (MEK inhibitor) for patients with metastatic non-small cell lung cancer (NSCLC) that was detected
More