By: Dr. Anish Shah Bronx-Lebanon Hospital; Bronx, NY On April 23, 2024, the U.S. Food and Drug Administration granted accelerated approval for tovorafenib (Ojemda, Day One Biopharmaceuticals, Inc.) for relapsed or refractory pediatric low-grade
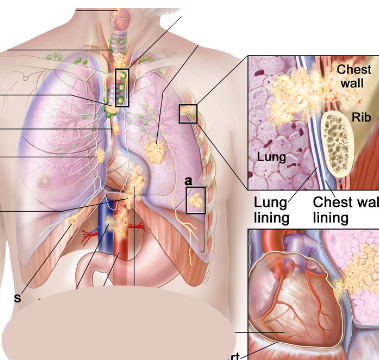
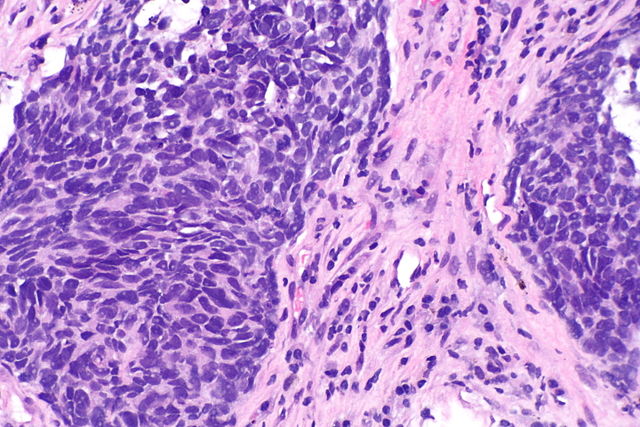
On October 11, 2023, the U.S. Food and Drug Administration granted approval to Encorafenib (BRAF inhibitor) with Binimetinib (MEK inhibitor) for patients with metastatic non-small cell lung cancer (NSCLC) that was detected
MoreAuthor: Saramshika Dhakal, MBBS Tribhuvan University Teaching Hospital, Kathmandu, Nepal. In a recent article published in JAMA Oncology (JAMA Oncol. 2023;9(4):465-472; researcher Simiao Chen and the team have estimated that the
MoreOn September 26, the U.S. Food and Drug Administration expanded the approval of bosutinib (Bosulif) to include pediatric patients 1 year of age and older. Previously, bosutinib had been approved for adults
MorePresented by: Chris Chen, MD, Director, Early Drug Development, Stanford Cancer Institute Covered by: Inas Abuali, MD, FACP Dr. Chris Chen, director of the Early Drug Development Program at Stanford Cancer Institute,
MoreOn August 28, 2023, the U.S. Food and Drug Administration approved luspatercept-aamt (Reblozyl) for the treatment of anemia in adult patients with very low- to intermediate-risk myelodysplastic syndromes (MDS) without prior erythropoiesis-stimulating agent (ESA) use,
MoreBy Akshat Jain MD, MPH Hematology Oncology and Cellular Therapy Faculty Department of Pediatrics & Clinical Medicine Loma Linda University School of Medicine Loma Linda University Children’s Hospital Assistant Professor of Public
More