On January 19, 2023, the U.S. Food and Drug Administration granted accelerated approval to Tucatinib plus Trastuzumab for adult patients with RAS wild-type metastatic colon cancer with HER2-positive status as detected at
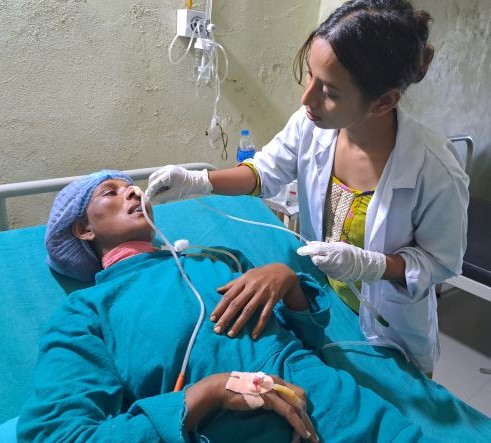
Dr. Rubina Suwal of the Binaytara Foundation Cancer Center performs successful oral cancer surgery for patient in local community
MoreThe Binaytara Foundation hosted two conferences in September 2022
MoreFall brings five conferences to the Pacific Northwest and Utah The Binaytara Foundation has organized five conferences in September and October, located in three states, Washington, Oregon, and Utah. Most of the
MoreASCO Direct™ Highlights Oncology conference held in Seattle to present and discuss the latest findings in cancer research and treatments The Binaytara Foundation held the ASCO Direct™ Highlights: 2022 Official Annual Meeting
MoreThe Binaytara Foundation (BTF) is pleased to welcome Dr. Sri Tantravahi as the new Co-Chair of the BTF Education Academy. Dr. Tantravahi will work closely with Dr. Sid Yadav, who will also
MoreAs part of our series of interviews with the 2022 Binaytara Foundation (BTF) award winners who are improving access to cancer care, today we talk with Dr. Bhawna Sirohi, winner of the
MoreIn this blog article, Dr. Dan Milner, Chief Medical Officer of the American Society for Clinical Pathology, discusses his experience about his participation in the 2022 Summit on Cancer Health Disparities held
MoreJody Stroh is the Binaytara Foundation's 2022 Volunteer of the Year.
MoreJanakpurdham, Nepal, April 17, 2022 – The Binaytara Cancer Trust (the non-profit organization that operates the Binaytara Foundation Cancer Center) held a Supporter’s Dinner on April 15 to thank the donors, supporters,
MoreJanakpurdham, Nepal, April 16, 2022 – The Binaytara Cancer Trust held a groundbreaking ceremony for the first phase of its planned 200-bed cancer hospital in Janakpurdham, Nepal on April 15. The Binaytara
More